News
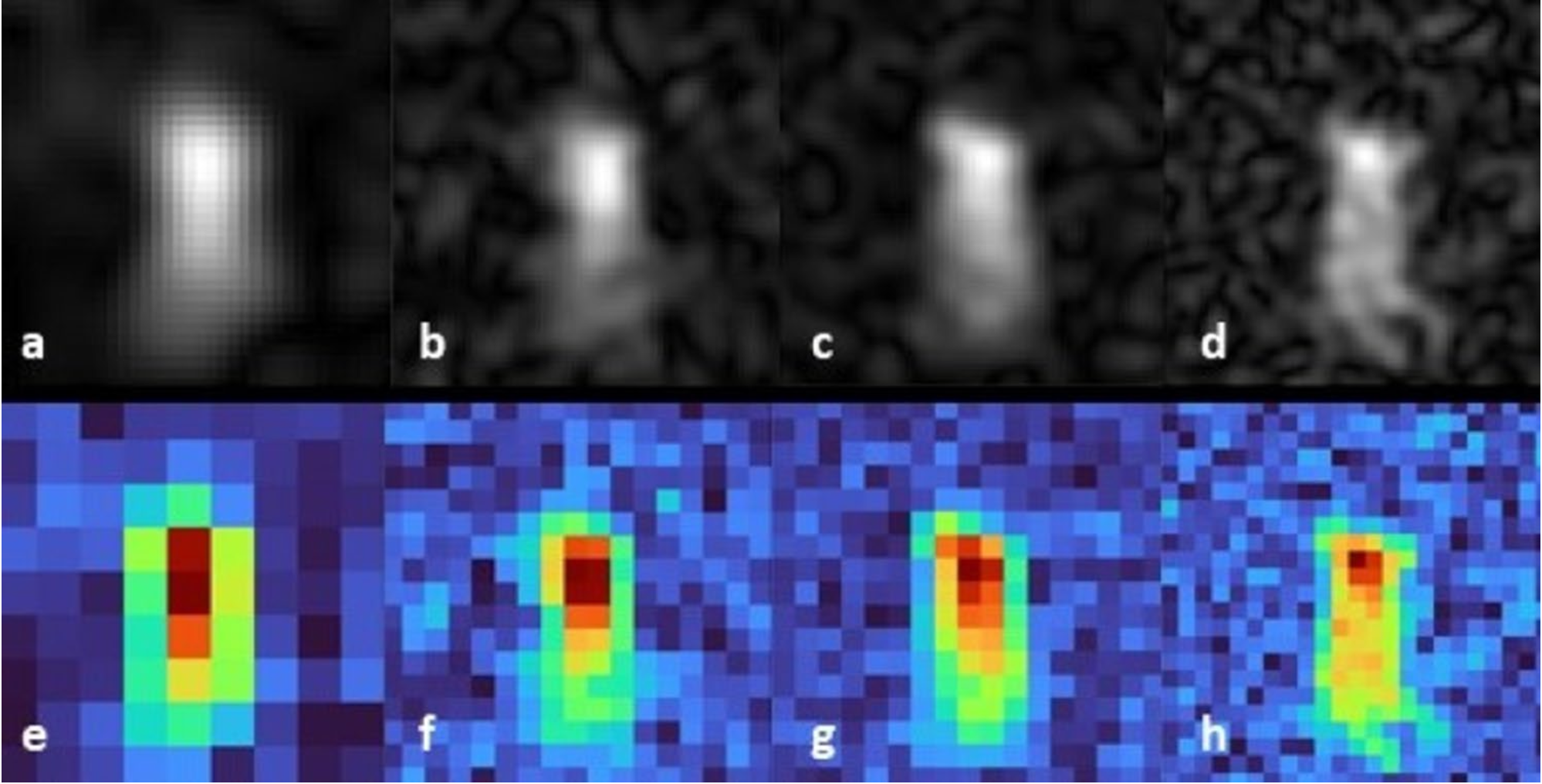
Publikation: "Detection of sub‐nmol amounts of the antiviral drug favipiravir in 19F MRI using photo‐chemically induced dynamic nuclear polarization"
Am 17. Januar wurde ein neuer Artikel in Scientific Reports veröffentlicht. Gratulation an die Autoren Johannes Bernarding, Christian Bruns, Isabell Prediger, Markus Mützel und Markus Plaumann.
Es ist gelungen kleinste Mengen Favipiravir mittels photo-CIDNP zu detektieren.
Hier geht es zu der Veröffentlichung: LINK
Detection of sub‐nmol amounts of the antiviral drug favipiravir in 19F MRI using photo‐chemically induced dynamic nuclear polarization
GCP courses in September 2023
The GCP course days (basic course and advanced course relating to the German Medicinal products Act AMG), which are held twice a year by the IBMI, took place on 11 and 12 September 2023. After the end of the coronavirus pandemic, they were once more held in person on Campus.
The participants included members of study teams as well as investigating physicians and study nurses. Both the GCP basic course (11 September) and the AMG advanced course (12 September) ended with an electronic multiple choice exam. All graduates of the course receive a certificate that entitles them to work on study teams and which is recognized by the Ethics Committees and the Higher Federal Authorities.
More information on the content of both course days can be found here.
As in previous years, we were able to rely on the tried and tested cooperation of pharmtrace klinische Entwicklung GmbH Berlin, the Coordination Center for Clinical Studies Magdeburg (KKS MD), the Ethics Committee of OVGU and the University Hospital Central Pharmacy (ZAP). We would also like to offer our warmest thanks to the representatives of these organizations, Dr. Richter (pharmtrace GmbH Berlin), Dr. Wiede (Coordination Center for Clinical Studies, KKS MD), Dr. Beck (Director of the Ethics Committee) and Dr. Kramer (Central Pharmacy, ZAP) for their expertise.
Publication: "Two fluorinated thulium complexes as molecular temperature sensors in MR application"
Congratulations to Felix Mysegaes, Isabell Prediger, Johannes Bernarding and Markus Plaumann, co-authors of "Two fluorinated thulium complexes as molecular temperature sensors in MR application" on the publication of their article in the journal ChemComm. They describe 19F-labeled complexes that allow temperature measurement by magnetic resonance spectroscopy.

DataDay: Insights into the World of Data Trustees
Data are essential for science and research. Data trust structures such as the data trust (THS) at IBMI come into play to protect them. At DataDay on 30 June 2023, our colleagues learned about and discussed opportunities for data trusteeship.
Speakers, such as our former intern Hanna Püschel, among others, provided insights into how legal scholars evaluate the idea of a data trust and how data intermediaries are used in medicine, among other fields.






